Hillrom and Welch Allyn are a part of Baxter.

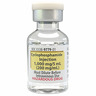
Cyclophosphamide Injection 1000 mg/5 mL (200 mg/mL)

Teilenr.: NVCP1000VUS
GTIN:
00303389779018
GTIN-CS:
50303389779013
Zum Warenkorb hinzugefügt
Produktverfügbarkeit nach Geschäftsstandort
Uhrzeiten
Cyclophosphamide Injection Solution 1000 mg/5 mL (200 mg/mL) in a multiple-dose vial. 1 vial per carton: NDC: 0338-9779-01
 Print
Print
 Email
Email
text.characteristics.heading
| text.partdetail.page.tradeMark | Baxter is trademark of Baxter International Inc. or its subsidiaries |
| text.characteristics.volume.name | 5.0 ml |
| text.characteristics.dehpFree.name | text.yes |
| text.characteristics.pvcFree.name | text.yes |
| text.characteristics.latexFree.name | text.yes |
| text.characteristics.shelfLifeFromManufacturer.name | 18 months |
| text.characteristics.concentration2.name | 200 mg/mL |
| text.characteristics.containerType.name | Glass Vial |
| text.characteristics.containesPreservative.name | text.yes |
| text.characteristics.dosageForm.name | Liquid |
| text.characteristics.polyethleneLined.name | text.no |
| text.characteristics.protectFromLight.name | text.no |
| text.characteristics.storageRecommendations.name | Store vial refrigerated at 2°C to 8°C (36°F to 46°F). After first use; store partially used multiple-dose vial in the original carton at 2°C to 8°C (36ºF to 46°F) for up to 28 days. Discard unused portion after 28 days. |
| text.characteristics.strength.name | 1000 mg/5 mL |
text.Carton.heading
| text.carton.unitsPerCase | 1.0 |
| text.carton.cartonWeight | 0.06 Pfund |
| text.carton.cartonLength | 1.78 Zoll |
| text.carton.cartonWidth | 1.53 Zoll |
| text.carton.cartonHeight | 3.56 Zoll |
| text.carton.cartonVolume | 0.01 fc |
text.hcpcs.heading
| text.hcpcs.code | text.hcpcs.description |
| J9076 | Injection, cyclophosphamide (baxter), 5 mg |
| text.hcpcsContactNo.name | 888-338-0001 |
text.hcpcs.informationSource.nametext.hcpcs.informationSource.cms
text.hcpcs.disclaimer.nametext.hcpcs.disclaimer.value
text.hcpcs.disclaimer.nametext.hcpcs.disclaimer.value
| text.orderingInformation.materialNumber.name | NVCP1000VUS |
| text.orderingInformation.unit.name | box |
| text.orderingInformation.ean.name | 00303389779018 |
| text.orderingInformation.eanCarton.name | 50303389779013 |
| text.orderingInformation.nationalProductCode.name | 0338977901 |
Andere
203052r1
Möchten Sie die Änderungen speichern, bevor Sie gehen?
Wir bearbeiten Ihre Anfrage. In dieser Zeit dürfen Sie das Fenster weder verlassen, noch aktualisieren oder schließen.
Bitte warten...
Bitte warten...